research use only
pomaglumetad (LY404039) GluR agonist
Cat.No.S6001
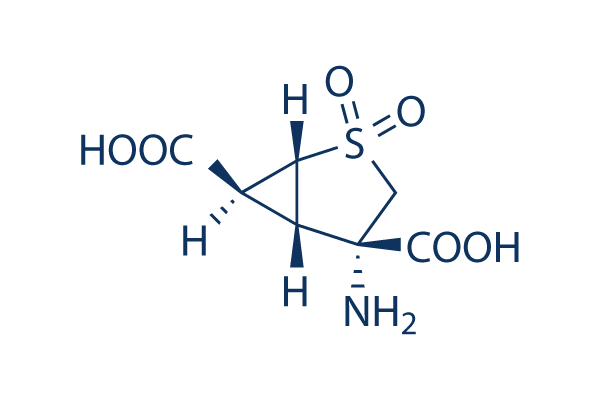
Chemical Structure
Molecular Weight: 235.22
Quality Control
Chemical Information, Storage & Stability
| Molecular Weight | 235.22 | Formula | C7H9NO6S |
Storage (From the date of receipt) | |
|---|---|---|---|---|---|
| CAS No. | 635318-11-5 | Download SDF | Storage of Stock Solutions |
|
|
| Synonyms | N/A | Smiles | C1C(C2C(C2S1(=O)=O)C(=O)O)(C(=O)O)N | ||
Solubility
|
In vitro |
5%TFA : 3.06 mg/mL
DMSO
: 0.5 mg/mL
(2.12 mM)
Water : Insoluble |
Molarity Calculator
|
In vivo |
|||||
In vivo Formulation Calculator (Clear solution)
Step 1: Enter information below (Recommended: An additional animal making an allowance for loss during the experiment)
Step 2: Enter the in vivo formulation (This is only the calculator, not formulation. Please contact us first if there is no in vivo formulation at the solubility Section.)
Calculation results:
Working concentration: mg/ml;
Method for preparing DMSO master liquid: mg drug pre-dissolved in μL DMSO ( Master liquid concentration mg/mL, Please contact us first if the concentration exceeds the DMSO solubility of the batch of drug. )
Method for preparing in vivo formulation: Take μL DMSO master liquid, next addμL PEG300, mix and clarify, next addμL Tween 80, mix and clarify, next add μL ddH2O, mix and clarify.
Method for preparing in vivo formulation: Take μL DMSO master liquid, next add μL Corn oil, mix and clarify.
Note: 1. Please make sure the liquid is clear before adding the next solvent.
2. Be sure to add the solvent(s) in order. You must ensure that the solution obtained, in the previous addition, is a clear solution before proceeding to add the next solvent. Physical methods such as vortex, ultrasound or hot water bath can be used to aid dissolving.
Mechanism of Action
| Features |
Under investigation as an exciting new medicine that may herald the arrival of third-generation antipsychotic drugs.
|
|---|---|
| Targets/IC50/Ki |
Rat neurons expressing native mGlu2/3
88 nM(Ki)
Recombinant human mGlu3
92 nM(Ki)
Recombinant human mGlu2
149 nM(Ki)
|
| In vitro |
Pomaglumetad (LY404039) exhibits low binding affinity to group III mGlu receptors including mGlu6, mGlu7 and mGlu8 with a Ki value more than 5 μM, and shows little affinity for ionotropic glutamate receptors, glutamate transporter subtypes, monoamine and other receptors. It potently inhibits forskolin-stimulated cAMP formation in cells expressing human mGlu2 and mGlu3 receptors. This compound suppresses electrically evoked excitatory activity in the striatum, and serotonin-induced L-glutamate release in the prefrontal cortex. It could modulate glutamatergic activity in limbic and forebrain areas relevant to psychiatric disorders and may be devoid of negative side effects associated with current antipsychotics and anxiolytics.
|
| In vivo |
Pomaglumetad (LY404039) demonstrates higher plasma exposure and better oral bioavailability, and may be valuable in the treatment of neuropsychiatric disorders, including anxiety and psychosis. In wild-type animals, this compound significantly reverses d-amphetamine(AMP)-induced increase in ambulations, distance travelled, and reduced time spent at rest. It reverses phencyclidine (PCP)-evoked behaviours at 10 mg/kg. The antipsychotic-like effects of LY404039 on PCP and AMP-evoked behavioural activation are absent in mGlu2 and mGlu2/3 but not in mGlu3 receptor-deficient mice. In contrast, clozapine and risperidone inhibit PCP-evoked behaviours in both wild-type and mGlu2/3 receptor-deficient mice. It reduces responding on the EtOH in the pavlovian spontaneous recovery (PSR) test and reduces the expression of an alcohol deprivation effect (ADE) during relapse, but does not affect EtOH responding under maintenance conditions. This compound inhibits the expression of alcohol seeking and relapse behaviour without altering alcohol self-administration behaviour. Moreover, it attenuates amphetamine- and phencyclidine-induced hyperlocomotion. It could inhibit conditioned avoidance responding and also reduces fear-potentiated startle in rats and marble burying in mice. Importantly, it does not produce sedative effects or motor impairment in the conditioned avoidance task. It also increases dopamine and serotonin release/turnover in the prefrontal cortex.
|
References |
|
Clinical Trial Information
(data from https://clinicaltrials.gov, updated on 2024-05-22)
| NCT Number | Recruitment | Conditions | Sponsor/Collaborators | Start Date | Phases |
|---|---|---|---|---|---|
| NCT01659177 | Withdrawn | Healthy Participants |
Denovo Biopharma LLC |
August 2012 | Phase 1 |
| NCT01637142 | Completed | Healthy Participants |
Denovo Biopharma LLC |
July 2012 | Phase 1 |
| NCT01609218 | Completed | Healthy Volunteer Study |
Denovo Biopharma LLC |
June 2012 | Phase 1 |
| NCT01475136 | Completed | Hepatic Insufficiency |
Denovo Biopharma LLC |
November 2011 | Phase 1 |
Tech Support
Tel: +1-832-582-8158 Ext:3
If you have any other enquiries, please leave a message.
